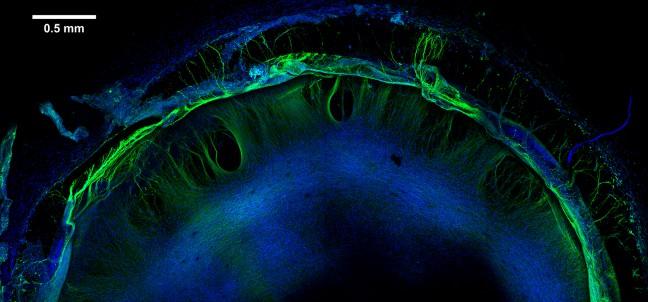
University of Wisconsin scientists have created a structure that mimics brain function and may be used to determine the safety of pharmaceutical drugs and industrial chemicals.
Using a hydrogel, a gel-like substance that readily absorbs water and stem cells, UW and Morgridge Institute for Research scientists have been able to successfully create cellular structures that mimic how brain tissue functions, Michael Schwartz, an assistant scientist in biomedical engineering at UW, said.
The program is called the “Tissue Chip” program, and in addition to UW, there are 10 other universities working on the project. The goal is to create various types of human tissues in order to create better methods of screening pharmaceutical drugs, William Murphy, co-director of the UW Stem Cell and Regenerative Medicine Center, said.
This research is part of a collaboration between the National Institutes of Health, The Defense Advanced Research Projects Agency and the Food and Drug Administration, Murphy said.
“The goal is to create various types of human tissues and then connect them to one another in a way that would be similar to how they connect in the body,” he said.
Scientists know very little about what chemicals are toxic to the human brain, and even less about how various chemicals affect a developing human brain, Schwartz said. Since tissues can’t grow independently, there was no way to study neurotoxicity in a lab. Much of the research done in the past was on those who had already been unwittingly affected by toxic chemicals, he said.
The new process will allow scientists to test drugs that are undergoing screening as well as industrial chemicals — many of which are never tested for toxicity on the developing human brain.
The structure created at UW is superior to previous structures because it is reproducible, Schwartz said. In past experiments, many of the tissues had to be kept in very specific conditions and would often not form the same way every time. In order for the tissues to work well as part of a drug testing process, all of the tissue samples needed to be uniform.
“Our procedure was developed to overcome this,” Schwartz said. “We are using a synthetic material that we know is chemically the same every single time, and we can make hundreds of tissues.”
In order to test their structure in terms of how well it can reveal neurotoxicity, Schwartz and his team gathered 70 different chemicals. Some of these chemicals were known to be toxic, and some were nontoxic. They used 60 of these chemicals to build their model, by exposing the tissue to them and then looking at the tissue’s responses.
They used the final 10 chemicals to test the accuracy of their model. Based on the response from the tissue, they were able to correctly determine the toxicity or nontoxicity of nine out of the 10 chemicals.
“We are very happily surprised. Our initial goal was 70 percent accuracy,” Schwartz said. “We never had any expectations one way or the other whether we’d be able to achieve that. We were shooting for 70 percent because that’s about the level of prediction that you can get if you use multiple animal studies.”
This type of drug testing could eventually appease those against the experimental use of animals by replacing animal testing during drug screening processes, Murphy said.
Though the initial testing has offered encouragement, the testing and development is far from complete, Schwartz said. Currently, the team is still working to validate this test as a model. Creating larger systems where different types of bodily tissue is connected would give scientists a better idea of how toxins affect the body holistically, Murphy said.
“These applications are really exciting and could be enabling in a variety of ways,” Murphy said. “There’s a whole lot we can understand by creating tissues in a dish.”